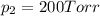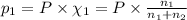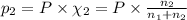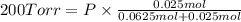Chemistry, 13.08.2019 02:30 lailahussain99
Agas mixture consists of equal masses of methane (molecular weight 16.0) and argon (atomic weight 40.0). if the partial pressure of argon is 200. torr, what is the pressure of methane, in torr? hint: what is the mole fraction of each gas?

Answers: 1
Another question on Chemistry



Chemistry, 22.06.2019 21:30
What is the effect of returning nuclear reactor cooling water back into bodies of water?
Answers: 3

You know the right answer?
Agas mixture consists of equal masses of methane (molecular weight 16.0) and argon (atomic weight 40...
Questions


Physics, 01.10.2019 20:10



Mathematics, 01.10.2019 20:10






Mathematics, 01.10.2019 20:10

English, 01.10.2019 20:10




History, 01.10.2019 20:10

Mathematics, 01.10.2019 20:10



History, 01.10.2019 20:10

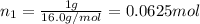
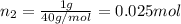
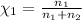
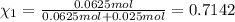
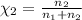
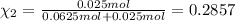
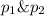 .
.