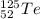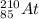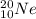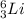Chemistry, 12.08.2019 19:30 pizzaqueen95
Write a symbol for each of the following neutral isotopes. include the atomic number and mass number for each.
(a) the chalcogen with a mass number of 125
(b) the halogen whose longest-lived isotope is radioactive
(c) the noble gas, used in lighting, with 10 electrons and 10 neutrons
(d) the lightest alkali metal with three neutrons

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:00
What layer of the atmosphere is directly above the troposphere?
Answers: 1

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 m koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 17:00
How can a give a full method for the experiment of separating sand from water by filtration? 1-materials 2-steps 3-conclusion also for water and salt separated by the evaporation or distillation process
Answers: 1

Chemistry, 22.06.2019 20:00
Carbon-14 undergoes radioactive decay in the reaction above. determine the type of radiation emitted in this reaction and describe what is happening to the nucleus during this reaction.
Answers: 2
You know the right answer?
Write a symbol for each of the following neutral isotopes. include the atomic number and mass number...
Questions

Mathematics, 02.04.2021 02:20

Mathematics, 02.04.2021 02:20

Social Studies, 02.04.2021 02:20


Mathematics, 02.04.2021 02:20

Mathematics, 02.04.2021 02:20

Mathematics, 02.04.2021 02:20


Mathematics, 02.04.2021 02:20


Mathematics, 02.04.2021 02:20

Mathematics, 02.04.2021 02:20


Social Studies, 02.04.2021 02:20


Mathematics, 02.04.2021 02:20

Social Studies, 02.04.2021 02:20


Mathematics, 02.04.2021 02:20

Mathematics, 02.04.2021 02:20