
Chemistry, 12.08.2019 18:30 briarkaltvedt
Calculate δ h° for the reaction c 4h 4( g) + 2h 2( g) → c 4h 8( g), using the following data: δ h° combustion for c 4h 4( g) = –2341 kj/mol δ h° combustion for h 2( g) = –286 kj/mol δ h° combustion for c 4h 8( g) = –2755 kj/mol

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 12:30
Write the chemical formula for a compound that is made of an element from group 1 and an element from group 17
Answers: 1

Chemistry, 22.06.2019 18:30
How many moles of bromine are needed to produce 3.23 moles of potassium bromide
Answers: 1

Chemistry, 23.06.2019 01:00
Which fossil fuel is mainly used for heating and cooking? a. electricity b. coal c. petroleum d. natural gas
Answers: 2

Chemistry, 23.06.2019 04:00
What are the names of these two interactions with cattle and how do they differ from each other
Answers: 3
You know the right answer?
Calculate δ h° for the reaction c 4h 4( g) + 2h 2( g) → c 4h 8( g), using the following data: δ h°...
Questions


Mathematics, 18.03.2021 03:10

History, 18.03.2021 03:10

History, 18.03.2021 03:10

History, 18.03.2021 03:10


Mathematics, 18.03.2021 03:10



Biology, 18.03.2021 03:10




Mathematics, 18.03.2021 03:10

Mathematics, 18.03.2021 03:10





Engineering, 18.03.2021 03:10

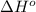
![\Delta H^o_{rxn}=\sum [n\times \Delta H^o_{(product)}]-\sum [n\times \Delta H^o_{(reactant)}]](/tpl/images/0174/3643/6872e.png)
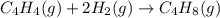
![\Delta H^o_{rxn}=[(1\times \Delta H^o_{(C_4H_8(g))})]-[(1\times \Delta H^o_{(C_4H_4(g))})+(2\times \Delta H^o_{(H_2(g))})]](/tpl/images/0174/3643/605f9.png)
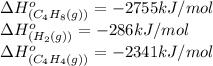
![\Delta H^o_{rxn}=[(1\times (-2755))]-[(1\times (-286))+(2\times (-2341))]\\\\\Delta H^o_{rxn}=2213kJ](/tpl/images/0174/3643/044fa.png)


