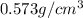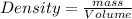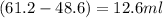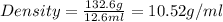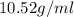Chemistry, 12.08.2019 17:20 jhanley4637
Alarge piece of jewelry has a mass of 132.6 g. a graduated cylinder initially contains 48.6 ml water. when the jewelry is submerged in the graduated cylinder, the total volume increases to 61.2 ml.
(a) determine the density of this piece of jewelry.
(b) assuming that the jewelry is made from only one suvstance, what substance is it likely to be? explain

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 23:00
The data below were determined for the reaction shown below. s2o82– + 3i – (aq) → 2so42– + i3– expt. # [s2o82–] (m) [i –] (m) initial rate 1 0.038 0.060 1.4 × 10 – 5 m/s 2 0.076 0.060 2.8 × 10 – 5 m/s 3 0.076 0.030 1.4 × 10 – 5 m/s the rate law for this reaction must be:
Answers: 1


Chemistry, 23.06.2019 03:00
In which of the following phases of matter do molecules have the highest amount of energy? a. liquid b. gel c. solid d. gas
Answers: 2

Chemistry, 23.06.2019 13:20
In the haber reaction, patented by german chemist fritz haber in 1908, dinitrogen gas combines with dihydrogen gas to produce gaseous ammonia. this reaction is now the first step taken to make most of the world's fertilizer. suppose a chemical engineer studying a new catalyst for the haber reaction finds that 671 liters per second of dinitrogen are consumed when the reaction is run at 271c and 0.99atm. calculate the rate at which ammonia is being produced. give your answer in kilograms per second. round your answer to significant digits.
Answers: 3
You know the right answer?
Alarge piece of jewelry has a mass of 132.6 g. a graduated cylinder initially contains 48.6 ml water...
Questions

Physics, 16.06.2021 14:00

English, 16.06.2021 14:00

Mathematics, 16.06.2021 14:00



Mathematics, 16.06.2021 14:00


Biology, 16.06.2021 14:00

Mathematics, 16.06.2021 14:00

Mathematics, 16.06.2021 14:00

Biology, 16.06.2021 14:00

Business, 16.06.2021 14:00

Social Studies, 16.06.2021 14:00

Engineering, 16.06.2021 14:00


Business, 16.06.2021 14:00

Mathematics, 16.06.2021 14:00

World Languages, 16.06.2021 14:00

Physics, 16.06.2021 14:00