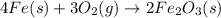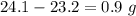Chemistry, 12.08.2019 16:20 novarosell
When elemental iron corrodes it combines with oxygen in the air to ultimately form red brown iron (iii) oxide which we call rust. (a) if a shiny ironnail with an initial mass of 23.2 g is weighed after being coated in a layer of rust, would you expect the mass to have increased, decreased, or remained the same? explain. (b) if the mass of the iron nall increases to 24.1 g what mass of oxygen combined with the iron?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 18:30
How much energy moves onto the next level, in an energy pyramid
Answers: 1

Chemistry, 21.06.2019 22:30
Which of these sequences lists the correct order for the creation of sedimentary rock from sediment? a. deposition, burial, compaction, cementation b. burial, deposition, compaction, cementation c. compaction, deposition, burial, cementation d. cementation, deposition, burial, compaction
Answers: 1

Chemistry, 22.06.2019 17:00
Astable electron arrangement for an atom is one that does not easily change. how is this arrangement arrived at? a. valence electrons are transferred or shared to create a full outer shell of electrons. b. valence electrons are discarded into space to create a full outer shell of electrons. c. protons (positive charge) pair with valence electrons (negative charge) to create a strong bond. d. outer shells with valence electrons are transferred or shared.
Answers: 2

Chemistry, 22.06.2019 17:30
Upon decomposition, one sample of magnesium fluoride produced 1.65 kg of magnesium and 2.56 kg of fluorine. a second sample produced 1.32 kg of magnesium. part a how much fluorine (in grams) did the second sample produce?
Answers: 2
You know the right answer?
When elemental iron corrodes it combines with oxygen in the air to ultimately form red brown iron (i...
Questions

History, 23.07.2019 18:00

Mathematics, 23.07.2019 18:00

History, 23.07.2019 18:00



English, 23.07.2019 18:00

Biology, 23.07.2019 18:00

Mathematics, 23.07.2019 18:00

Mathematics, 23.07.2019 18:00

Physics, 23.07.2019 18:00


SAT, 23.07.2019 18:00

Mathematics, 23.07.2019 18:00




Spanish, 23.07.2019 18:00

Biology, 23.07.2019 18:00

History, 23.07.2019 18:00