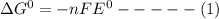Consider the following half reaction in which nitrate is reduced to nitrite: no3– 2h 2e– → no2– h2o. the reaction has a δg°' value of –81 kj/mol. the negative value of δg°' results from the very e°' value of the redox couple no3–/no2–, and it reflects the strong tendency of nitrate to electrons. choose one: a. positive / donateb. negative/ acceptc. negative / donated. positive / accept

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 14:30
How many joules of heat are absorbed to raise the temperature of 650 grams of water from 5.00c to it's boiling point, 100c
Answers: 1

Chemistry, 21.06.2019 18:30
Strong conductivity of plasma allows it to act and react as and
Answers: 2

Chemistry, 22.06.2019 05:30
You are making a solution of calcium chloride dissolved in water. you add solid, stir, and it dissolves. you add just a spatula tip full, stir, and the solid does not dissolve. how could you describe the solutions before and after adding the spatula tip amount
Answers: 1

Chemistry, 22.06.2019 06:00
An alkaline battery produces electrical energy according to the following equation. zn(s) + 2 mno2(s) + h2o(l) zn(oh)2(s) + mn2o3(s) (a) determine the limiting reactant if 17.5 g zn and 31.0 g mno2 are used. (type your answer using the format ch4 for ch4.) (b) determine the mass of zn(oh)2 produced. _ g
Answers: 3
You know the right answer?
Consider the following half reaction in which nitrate is reduced to nitrite: no3– 2h 2e– → no2– h2o...
Questions



Physics, 01.11.2020 01:00



Mathematics, 01.11.2020 01:00

Computers and Technology, 01.11.2020 01:00




Biology, 01.11.2020 01:00

Mathematics, 01.11.2020 01:00

Mathematics, 01.11.2020 01:00

Biology, 01.11.2020 01:00




Arts, 01.11.2020 01:00

English, 01.11.2020 01:00