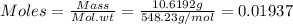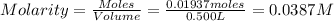Astandard solution is prepared by dissolving 10.6192 g of (nh4)2ce(no3)6 (548.23 g•mol-1, 98.75% purity) in dilute sulfuric acid. the resulting solution is quantitatively transferred to a 500.0-ml volumetric flask and diluted to the mark. what is the ce concentration in the final solution?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 16:30
What is the force of attraction between the particles in a salt crystal
Answers: 2

Chemistry, 22.06.2019 00:20
Use the gizmo to find the concentration of the mystery ch3cooh. use the titrant and indicator shown below perform the titration. what is the titrant volume? titrant analyte indicator titrant volume analyte concentration naoh ch3cooh phenophthalein select one: a. 20.0 ml b. 27.0 ml c. 30.0 ml d. 24.0 ml
Answers: 2

Chemistry, 22.06.2019 08:30
Which part of earth’s surface receives the most direct rays from the sun? a) equator b) ocean c) poles d) mountains
Answers: 2

Chemistry, 22.06.2019 19:10
Astudent completes a titration by adding 12.0 milliliters of naoh(aq) of unknown concentration to 16.0 milliliters of 0.15 m hcl(aq). what is the molar concentration of the naoh(aq)? 1)5.0 m 2)0.20 m 3)0.11 m 4)1.1 m
Answers: 1
You know the right answer?
Astandard solution is prepared by dissolving 10.6192 g of (nh4)2ce(no3)6 (548.23 g•mol-1, 98.75% pur...
Questions




Health, 18.08.2019 19:30


Chemistry, 18.08.2019 19:30

Biology, 18.08.2019 19:30

Mathematics, 18.08.2019 19:30


History, 18.08.2019 19:30

Social Studies, 18.08.2019 19:30

Mathematics, 18.08.2019 19:30


Chemistry, 18.08.2019 19:30

History, 18.08.2019 19:30



Biology, 18.08.2019 19:30


Mathematics, 18.08.2019 19:30