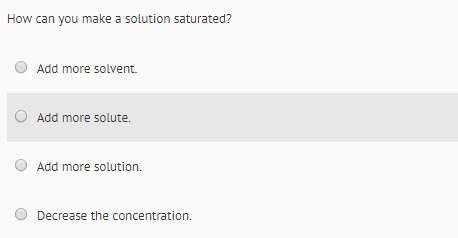
Consider the following reaction mechanism:
step 1: (ch3)3auph3 h(ch3)3au + ph3 (fast)
...

Chemistry, 09.08.2019 01:30 hwilson118027
Consider the following reaction mechanism:
step 1: (ch3)3auph3 h(ch3)3au + ph3 (fast)
step 2: (ch3)3au → c2h6 + (ch3)au (slow)
step 3: (ch3)au + ph3 → (ch3)auph3 (fast)
what is the balanced equation for the overall reaction?
o (ch3)3auph3 → c2h6 + (ch3)auph3
(ch3)3auph3 → c2h6 + (ch3)au + ph3
(ch3)3auph3 → (ch3)au + ph3
o (ch3)3auph3 – ch3 + (c2h6)au + ph3

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:00
For ai it's atomic number is 13 and it's mass number is 27 how many neutrons does it have
Answers: 1

Chemistry, 23.06.2019 02:30
what is your question? collegechemistry 5+3 pts in november 1987, a massive iceberg broke loose from the antartic ice mass and floated free in the ocean. the chunk of ice was estimated to be 98 mi long, 25 mi wide, and 750 ft thick. a typical backyard swimming pool contains about 24,000 gallons of water. how many of these pools could you fill from the water in this iceberg? (assume the iceberg is a rectangular solid of the above dimensions and consists of water only). express answer in scientific notation.
Answers: 3

Chemistry, 23.06.2019 04:30
Two liquids are poured into a beaker. after a few seconds, the beaker becomes warm. which of the following best describes this reaction? a. an exothermic reaction b. a decomposition reaction c. an endothermic reaction d. a single-displacement reaction
Answers: 1

Chemistry, 23.06.2019 10:10
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10-3 m and k for the dissociation is 1.86x10-5. ch3cooh(aq)+h2o(l)+> h3o+(aq)+ch3coo-(aq) show me how to get the answer.
Answers: 3
You know the right answer?
Questions

History, 24.07.2019 17:20


Geography, 24.07.2019 17:20



History, 24.07.2019 17:20




Mathematics, 24.07.2019 17:20

Spanish, 24.07.2019 17:20

Mathematics, 24.07.2019 17:20




Mathematics, 24.07.2019 17:20


Geography, 24.07.2019 17:20

Mathematics, 24.07.2019 17:20

Social Studies, 24.07.2019 17:30