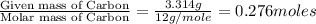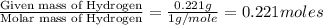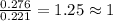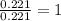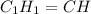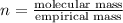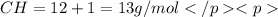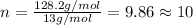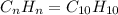Chemistry, 08.08.2019 06:20 alwaysneedhelp84
when 3.539 grams of a hydrocarbon, cxhy, were burned in a combustion analysis apparatus, 12.15 grams of co2 and 1.990 grams of h2o were produced.
in a separate experiment, the molar mass of the compound was found to be 128.2 g/mol. determine the empirical formula and the molecular formula of the hydrocarbon.
enter the elements in the order presented in the question.
empirical formula =
molecular formula =

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:30
Which statement best describes the relationship between period and frequency of light waves? a) in wave b the period increases and the frequency decreases from wave a. b) in wave a the period increases and the frequency decreases from wave b. c) in wave b the period is shorter and the frequency is greater than in wave a. d) in wave a the period is shorter and the frequency is greater than in wave b.
Answers: 1


Chemistry, 22.06.2019 16:50
Which element is least likely to undergo a chemical reaction
Answers: 3

You know the right answer?
when 3.539 grams of a hydrocarbon, cxhy, were burned in a combustion analysis apparatus, 12.15 grams...
Questions





History, 05.11.2020 21:20

English, 05.11.2020 21:20

Chemistry, 05.11.2020 21:20


English, 05.11.2020 21:20

Mathematics, 05.11.2020 21:20


Mathematics, 05.11.2020 21:20


Mathematics, 05.11.2020 21:20

Social Studies, 05.11.2020 21:20


Mathematics, 05.11.2020 21:20



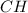 and
and 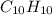 respectively.
respectively.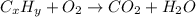
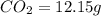
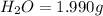
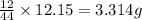 of carbon will be contained.
of carbon will be contained.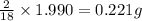 of hydrogen will be contained.
of hydrogen will be contained.