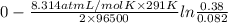Aconcentration cell is built based on the following half reactions by using two pieces of zinc as electrodes, two zn2+ solutions, 0.129 m and 0.427 m, and all other materials needed for a galvanic cell. what will the potential of this cell be when the cathode concentration of zn2+ has changed by 0.047 m at 291 k?
zn2+ + 2 e- ? zn eo = -0.761 v

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:00
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change
Answers: 3

Chemistry, 22.06.2019 19:50
Identify the lewis base in this balanced equation: fe3+ h2o fe(h2o)63+
Answers: 1


Chemistry, 23.06.2019 02:00
Why does ammonia, nh3, behave as a base when it reacts with an acid? z
Answers: 2
You know the right answer?
Aconcentration cell is built based on the following half reactions by using two pieces of zinc as el...
Questions

Mathematics, 17.01.2020 01:31

Mathematics, 17.01.2020 01:31

Mathematics, 17.01.2020 01:31

Mathematics, 17.01.2020 01:31


Mathematics, 17.01.2020 01:31


Mathematics, 17.01.2020 01:31


Advanced Placement (AP), 17.01.2020 01:31

English, 17.01.2020 01:31

Mathematics, 17.01.2020 01:31



Chemistry, 17.01.2020 01:31

Biology, 17.01.2020 01:31

Mathematics, 17.01.2020 01:31

Mathematics, 17.01.2020 01:31


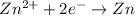 ,
, 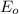 = -0.761 V
= -0.761 V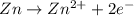 ,
, 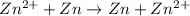
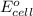 for the given reaction is zero.
for the given reaction is zero.![E^{o}_{cell} - \frac{RT}{nF} ln \frac{[Zn^{2+}]_{products}}{[Zn^{2+}]_{reactants}}](/tpl/images/0173/1699/4d9c9.png)