
Chemistry, 07.08.2019 03:10 Jazminnexoxo1093
The following thermodynamic data are available for octane, oxygen gas, carbon dioxide gas, water, and water vapor: molecule δh∘f (kj/mol) c8h18(l) −250.1 o2(g) 0 co2(g) −393.5 h2o(l) −285.8 h2o(g) −241.8 part b calculate δhrxn for the combustion of octane by using enthalpies of formation from the transition above. express the energy in kilojoules per mole to three significant figures. δhrxn δ h r x n = nothing kj/mol

Answers: 3
Another question on Chemistry



Chemistry, 23.06.2019 08:40
Which statement is true according to the kinetic theory? a. molecules of different gases with the same mass and temperature always have the same average density. b. molecules of different gases with the same mass and temperature always have the same average volume. c. molecules of different gases with the same mass and temperature always have the same pressure. d. molecules of different gases with the same mass and temperature always have the same molecular mass. e. molecules of different gases with the same mass and temperature always have the same average kinetic energy.
Answers: 1

Chemistry, 23.06.2019 18:30
Which of the following describes a base? select all that apply. a. tastes sour b. tastes bitter c. highly reactive d. slippery feel
Answers: 2
You know the right answer?
The following thermodynamic data are available for octane, oxygen gas, carbon dioxide gas, water, an...
Questions




Mathematics, 29.09.2020 06:01

Biology, 29.09.2020 06:01

Mathematics, 29.09.2020 06:01

History, 29.09.2020 06:01





Geography, 29.09.2020 06:01

Mathematics, 29.09.2020 06:01


Social Studies, 29.09.2020 06:01

Mathematics, 29.09.2020 06:01


Mathematics, 29.09.2020 06:01

Mathematics, 29.09.2020 06:01

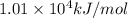
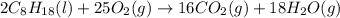
![\Delta H^o_{rxn}=\sum [n\times \Delta H^o_f(product)]-\sum [n\times \Delta H^o_f(reactant)]](/tpl/images/0172/5689/45485.png)
![\Delta H^o_{rxn}=[(16\times \Delta H^o_f_{(CO_2(g))})+(18\times \Delta H^o_f_{(H_2O(g))})]-[(2\times \Delta H^o_f_{(C_8H_{18}(l))})+(25\times \Delta H^o_f_{(O_2(g))})]](/tpl/images/0172/5689/36b5a.png)
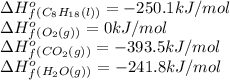
![\Delta H^o_{rxn}=[(16\times (-393.5))+(18\times (-241.8))]-[(2\times (-250.1))+(25\times (0))]=10148.2kJ/mol=1.01\times 10^4kJ/mol](/tpl/images/0172/5689/a24a9.png)


