
At 700 k, the reaction2so2(g) + o2(g) 2so3(g)has the equilibrium constant kc = 4.3 x 106, and the following concentrations are present: [so2] = 0.010 m; [so3] = 10.m; [o2] = 0.010 m. is the mixture at equilibrium? if not at equilibrium, in which direction (as the equation is written), left to right or right to left, will the reaction proceed to reach equilibrium? yes, the mixture is at equilibrium. no, left to right. no, right to left. there is not enough information to be able to predict the direction.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:40
Which of the following pressures is equal to 760 mm hg? 2.0 atm 101.3 pa 101,300 kpa 101,300 pa
Answers: 2

Chemistry, 22.06.2019 04:30
In which phase(s) do the molecules take the shape of the container?
Answers: 1

Chemistry, 22.06.2019 17:30
Oil rich countries in the middle east cover about 4% of earths total land area but prossess about 48% of the worlds known oil reserves what is the main reason for high concentration of reserves in this part of the world
Answers: 3

Chemistry, 22.06.2019 22:30
Astudent pours 10.0 g of salt into a container of water and observes the amount of time it takes for the salt to dissolve. she then repeats the process using the same amounts of salt and water but this time she slowly stirs the mixture while it is dissolving. the student performs the experiment one more time but this time she stirs the mixture rapidly. the dependent variable in this experiment is: time for salt to dissolve speed of stirring amount of water mass of salt
Answers: 1
You know the right answer?
At 700 k, the reaction2so2(g) + o2(g) 2so3(g)has the equilibrium constant kc = 4.3 x 106, and the fo...
Questions

Computers and Technology, 18.11.2020 01:40



Biology, 18.11.2020 01:40



Mathematics, 18.11.2020 01:40



Advanced Placement (AP), 18.11.2020 01:40

English, 18.11.2020 01:40



German, 18.11.2020 01:40

Mathematics, 18.11.2020 01:40



Biology, 18.11.2020 01:40


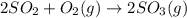
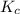 = 4.3 \times 10^{6}[/tex].
= 4.3 \times 10^{6}[/tex].![[SO_2]](/tpl/images/0172/4735/0303a.png) = 0.010 M;
= 0.010 M; ![[SO_3]](/tpl/images/0172/4735/8a06f.png) = 10.M;
= 10.M; ![[O_2]](/tpl/images/0172/4735/b0db0.png) = 0.010 M.
= 0.010 M.![\frac{[SO_{3}]^{2}}{[SO_{2}]^{2}[O_{2}]}](/tpl/images/0172/4735/2750c.png)
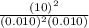
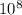
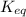 , then reaction moves in the backward direction.
, then reaction moves in the backward direction.

