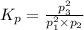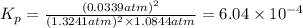Chemistry, 06.08.2019 05:10 alexsk6357
What is the value of kp for the reaction of nitrogen and oxygen to make dinitrogen monoxide if the equilibrium partial pressures of nitrogen is 1.3241 atm, the partial pressure of oxygen is 1.0844 atm and the partial pressure of dinitrogen monoxide is 0.0339 atm?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Why are the trends and exceptions to the trends in ionization energy observed?
Answers: 1

Chemistry, 22.06.2019 01:30
When an object falls through the air and encounters air resistance its overall speed will be than if it had not encountered air resistance? (one word answer)
Answers: 2

Chemistry, 22.06.2019 04:00
The continuous release of nuclear energy caused when one fission reaction triggered more nuclear reactions is a
Answers: 3

You know the right answer?
What is the value of kp for the reaction of nitrogen and oxygen to make dinitrogen monoxide if the e...
Questions



SAT, 30.11.2021 01:50




SAT, 30.11.2021 01:50

Mathematics, 30.11.2021 01:50


Computers and Technology, 30.11.2021 01:50






Computers and Technology, 30.11.2021 01:50

Health, 30.11.2021 01:50

Computers and Technology, 30.11.2021 01:50


Social Studies, 30.11.2021 01:50

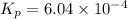
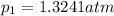
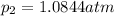
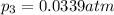
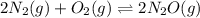
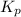 will be given as:
will be given as: