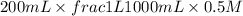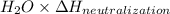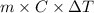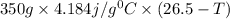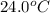Chemistry, 05.08.2019 19:10 camrenp9889
When 150. ml of 0.400 m h+ are mixed with 200. ml of 0.500 m oh-, the final temperature of the solution is 26.5°c. what was the initial temperature of the solution before the reaction occurred? assume that the solution has a total mass of 350. g and a specific heat capacity of 4.184 j/g°c. the enthalpy of neutralization for the reaction is -62.0 kj/mol of water produced.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 14:30
For the reaction shown, find the limiting reactant for each of the following initial amounts of reactants. 4al(s)+3o2(g)→2al2o3(s) a) 1 molal, 1 mol o2 b) 4 molal, 2.6 mol o2 c) 16 molal, 13 mol o2 d) 7.4 molal, 6.5 mol o2
Answers: 3

Chemistry, 23.06.2019 01:10
Can someone check my work 98 5.05 acids and bases for this assignment you will be comparing acids and bases. the chart below will you organize the information needed: acids bases chemical properties (2) deodorant detergent vinger dish soap physical properties (2) orange juice toilet cleaner drain cleaner window cleaner ph level acid ph goes from 0-4 bases ph goes from 10-14 examples around you (2) vinger coffee lemon juice dark chocolate
Answers: 3

Chemistry, 23.06.2019 04:31
Pls i will do pls imma diewhat forms white light? (4 points)a. combination of all wavelengths of ultraviolet light b. combination of all wavelengths of visible lightc. absorption of electromagnetic waves d. absorption of infrared rays
Answers: 2

Chemistry, 23.06.2019 08:30
Sand is more likely than shale to preserve fossils. true false
Answers: 2
You know the right answer?
When 150. ml of 0.400 m h+ are mixed with 200. ml of 0.500 m oh-, the final temperature of the solut...
Questions


Geography, 27.07.2019 05:30




Arts, 27.07.2019 05:30

Mathematics, 27.07.2019 05:30

Mathematics, 27.07.2019 05:30








History, 27.07.2019 05:30

History, 27.07.2019 05:30



Physics, 27.07.2019 05:30

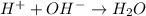
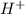 = volume × concentration of
= volume × concentration of 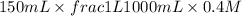
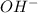 = volume × concentration of
= volume × concentration of