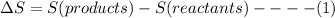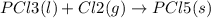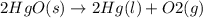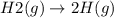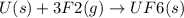Chemistry, 01.08.2019 05:10 nicole10perez
State whether the sign of the entropy change expected for each of the following processes will be positive or negative, and explain your predictions. (a) pcl3(l) + cl2(g) ⟶pcl5(s) (b) 2hgo(s) ⟶2hg(l) + o2(g) (c) h2(g) ⟶2h(g) (d) u(s) + 3f2(g) ⟶uf6(s)

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 07:30
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 3

Chemistry, 22.06.2019 17:30
Which scenario is most similar to the type of collision that gas particles have according to kinetic molecular theory
Answers: 1

Chemistry, 22.06.2019 21:00
Read "who built the pyramids? ”. leave this link open while you answer the questions throughout the assignment. give at least two reasons why some people claim the pyramids of giza were constructed by aliens.
Answers: 1
You know the right answer?
State whether the sign of the entropy change expected for each of the following processes will be po...
Questions


English, 14.12.2020 01:30

Mathematics, 14.12.2020 01:30

Computers and Technology, 14.12.2020 01:30



Mathematics, 14.12.2020 01:30

Mathematics, 14.12.2020 01:30


Mathematics, 14.12.2020 01:30










Mathematics, 14.12.2020 01:30