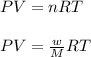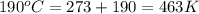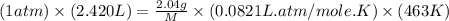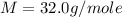Chemistry, 31.07.2019 21:20 rashawng2005
Asample of an unknown compound is vaporized at 190.°c. the gas produced has a volume of 2420.ml at a pressure of 1.00 atm, and it weighs 2.04 g. assuming the gas behaves as an ideal gas under these conditions, calculate the molar mass of the compound. be sure your answer has the correct number of significant digits.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:00
Particles vibrate in a rigid structure and do not move relative to their neighbors.
Answers: 1

Chemistry, 22.06.2019 12:00
What is the percentage of hydrogen in nitrogen trihydride
Answers: 1

Chemistry, 22.06.2019 14:40
Choose an equation that represents an enzyme-catalyzed reaction. (a) enzyme + substrate → enzyme-substrate complex (b) enzyme + substrate ←−→ enzyme + products (c) enzyme + substrate ←−→ enzyme-substrate complex → enzyme + products (d) enzyme + substrate ←−→ enzyme-substrate complex → enzyme-substrate complex + products
Answers: 2

Chemistry, 22.06.2019 16:00
What rule is used to determine how many covalent bonds an element can form? a. the number of covalent bonds is equal to six c the number of covalent bonds is equal to five minus the group number plus the group number b. the number of covalent bonds is equal to eight d. none of the above minus the group number select the best answer from the choices provided
Answers: 2
You know the right answer?
Asample of an unknown compound is vaporized at 190.°c. the gas produced has a volume of 2420.ml at a...
Questions

Mathematics, 23.02.2021 04:20




Mathematics, 23.02.2021 04:20

History, 23.02.2021 04:20



Mathematics, 23.02.2021 04:20

Social Studies, 23.02.2021 04:20



Chemistry, 23.02.2021 04:20

English, 23.02.2021 04:20


Mathematics, 23.02.2021 04:20

Mathematics, 23.02.2021 04:20

Mathematics, 23.02.2021 04:20

Spanish, 23.02.2021 04:20

Mathematics, 23.02.2021 04:20