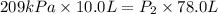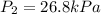Chemistry, 31.07.2019 17:20 houtchhaytang
Acylinder is filled with 10.0l of gas and a piston is put into it. the initial pressure of the gas is measured to be 209.kpa. the piston is now pulled up, expanding the gas, until the gas has a final volume of 78.0l. calculate the final pressure of the gas. be sure your answer has the correct number of significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
1.aluminum chloride (alcl3), and sodium hydroxide (naoh) can react to form aluminum hydroxide (al(oh)3) and sodium chloride (nacl). you have 13.4 g of aluminum chloride and 10.0 g of sodium hydroxide. answer the following questions: •what is the balanced equation for this reaction? •if you use all 13.4 g of aluminum chloride, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •if you use all 10.0 g of sodium hydroxide, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •how many grams of aluminum hydroxide will actually be made? which reagent is limiting? explain your answer.
Answers: 1

Chemistry, 22.06.2019 04:20
Which of the following is true for the actual yield of a reaction? it is always calculated as a ratio. it is the yield from the excess reactant. it is the yield from the limiting reactant. it is always less than the theoretical yield.
Answers: 1

Chemistry, 22.06.2019 08:30
Analyze how limestone is weathered and identify the features that are formed as a result of this dissolution
Answers: 1

Chemistry, 22.06.2019 10:30
When the speed of the bottle is 2 m/s, the average maximum height of the beanbag is m.
Answers: 2
You know the right answer?
Acylinder is filled with 10.0l of gas and a piston is put into it. the initial pressure of the gas i...
Questions




Social Studies, 06.10.2019 07:00


Social Studies, 06.10.2019 07:00


Mathematics, 06.10.2019 07:00

History, 06.10.2019 07:00

Mathematics, 06.10.2019 07:00





Mathematics, 06.10.2019 07:00

Mathematics, 06.10.2019 07:00

English, 06.10.2019 07:00



English, 06.10.2019 07:00

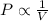
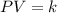
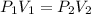
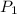 = initial pressure of the gas = 209 kPa
= initial pressure of the gas = 209 kPa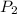 = final pressure of the gas = ?
= final pressure of the gas = ?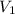 = initial volume of the gas = 10.0 L
= initial volume of the gas = 10.0 L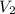 = final volume of the gas = 78.0 L
= final volume of the gas = 78.0 L