Chemistry, 30.07.2019 00:20 saabrrinnaaa
At 298 k, kc = 1.45 for the following reaction 2 brcl (g) br2(g) + cl2(g) a reaction mixture was prepared with the following initial concentrations. [brcl] = 0.0400 m, [br2] = 0.0300 m and [cl2] = 0.0300 m calculate their equilibrium concentrations.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:40
Kc = 0.040 for the system below at 450oc. if a reaction is initiated with 0.40 mole of cl2 and 0.40 mole of pcl3 in a 2.0 liter container, what is the equilibrium concentration of cl2 in the same system? pcl5(g) ⇄ pcl3(g) + cl2(g)
Answers: 3

Chemistry, 22.06.2019 10:10
What shape would a molecule with two bound groups and two lone pairs have?
Answers: 1

Chemistry, 22.06.2019 19:20
Consider hydrogen in an excited state n = 5n=5 that emits photons to reach the ground state. there are various possible transitions other than straight to the ground state that can occur; for example, it can drop to the n = 3n=3 state followed by the n = 3n=3 to the ground state transition. which of the possible transitions will result in the emission of a photon in the visible region?
Answers: 3

Chemistry, 22.06.2019 20:00
The picture represents the process that produces most of the energy used by living organisms on earth. which process is represented in the picture? a) the magnetic attraction between two hydrogen nuclei. b) the fusion of hydrogen nuclei to produce a helium nucleus in the core of the sun. c) the fission of hydrogen nuclei to produce a helium nucleus in the core of the sun. d) the chemical reaction between hydrogen nuclei to produce a helium nucleus in earth's atmosphere.
Answers: 3
You know the right answer?
At 298 k, kc = 1.45 for the following reaction 2 brcl (g) br2(g) + cl2(g) a reaction mixture was pre...
Questions

Mathematics, 02.11.2020 02:00

Mathematics, 02.11.2020 02:00

Mathematics, 02.11.2020 02:00



Mathematics, 02.11.2020 02:00

English, 02.11.2020 02:00


Mathematics, 02.11.2020 02:00

Mathematics, 02.11.2020 02:00

History, 02.11.2020 02:00

Mathematics, 02.11.2020 02:00



Physics, 02.11.2020 02:00

Mathematics, 02.11.2020 02:00


Chemistry, 02.11.2020 02:00


![1.5=K_{c}=\frac{[Br_{2}][Cl_{2}]}{[BrCl]^{2}}](/tpl/images/0148/5536/4f48c.png)
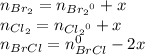
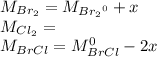

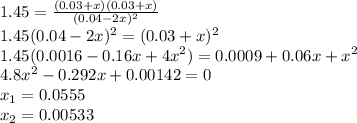
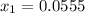 lacks of sense because it will give a negative concentration for BrCl, so the result is
lacks of sense because it will give a negative concentration for BrCl, so the result is 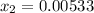 .
.