
Chemistry, 26.07.2019 20:30 joeblaszak4776
Three kilograms of steam is contained in a horizontal, frictionless piston and the cylinder is heated at a constant pressure of 0.5 bar from 100 °c to such a temperature that the specific volume increases by 2.5 times. if the amount of heat that must be added to accomplish this change is 500 kj, calculate the final temperature of the steam, the expansion work, and the change in internal energy.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 18:00
Aballoon inflated with three breaths of air has a volume of 1.7 l. at the same temperature and pressure, what is the volume of the balloon if five more same-sized breaths are added to the balloon? a balloon inflated with three breaths of air has a volume of 1.7 l. at the same temperature and pressure, what is the volume of the balloon if five more same-sized breaths are added to the balloon?
Answers: 3

Chemistry, 22.06.2019 15:00
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 1

Chemistry, 22.06.2019 19:30
Chlorine and water react to form hydrogen chloride and oxygen, like this: 2cl2 (g) + 2h2o (g) → 4hcl (g) + o2 (g) also, a chemist finds that at a certain temperature the equilibrium mixture of chlorine, water, hydrogen chloride, and oxygen has the following composition: compound concentration at equilibrium cl2 0.55m h2o 0.57m hcl 0.53m o2 0.34m calculate the value of the equilibrium constant kc for this reaction. round your answer to 2 significant digits.
Answers: 2

Chemistry, 23.06.2019 01:30
Which is an example of a highly unstable isotope that is often used in fission reactions?
Answers: 1
You know the right answer?
Three kilograms of steam is contained in a horizontal, frictionless piston and the cylinder is heate...
Questions


Mathematics, 08.11.2019 18:31

Mathematics, 08.11.2019 18:31

Advanced Placement (AP), 08.11.2019 18:31

Mathematics, 08.11.2019 18:31





History, 08.11.2019 18:31

English, 08.11.2019 18:31



Biology, 08.11.2019 18:31

Mathematics, 08.11.2019 18:31

History, 08.11.2019 18:31




English, 08.11.2019 18:31

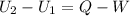
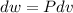
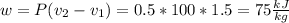 (There is a multiplication by 100 due to the conversion of bar to kPa)
(There is a multiplication by 100 due to the conversion of bar to kPa)
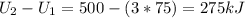
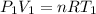
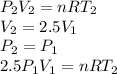
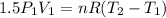
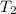 :
: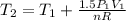
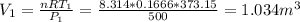
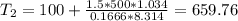 ºC
ºC

