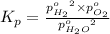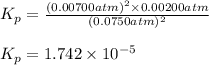Chemistry, 25.07.2019 01:20 jaymee2904p88tgh
The elementary reaction 2h2o(g)↽−−⇀2h2(g)+o2(g) proceeds at a certain temperature until the partial pressures of h2o, h2, and o2 reach 0.0750 atm, 0.00700 atm, and 0.00200 atm, respectively. what is the value of the equilibrium constant at this temperature?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:30
Which statement best describes the relationship between period and frequency of light waves? a) in wave b the period increases and the frequency decreases from wave a. b) in wave a the period increases and the frequency decreases from wave b. c) in wave b the period is shorter and the frequency is greater than in wave a. d) in wave a the period is shorter and the frequency is greater than in wave b.
Answers: 1

Chemistry, 22.06.2019 17:00
The atoms of a solid aluminum can are close together, vibrating in a rigid structure. if the can is warmed up on a hot plate, what happens to the atoms?
Answers: 1

Chemistry, 22.06.2019 17:30
Which scenario is most similar to the type of collision that gas particles have according to kinetic molecular theory
Answers: 1

Chemistry, 23.06.2019 01:00
An unsaturated hydrocarbon is a hydrogen-carbon compound with a. a network solid structure b. single bonds c. single bonds in a branched-chain structure d. double or triple bonds
Answers: 1
You know the right answer?
The elementary reaction 2h2o(g)↽−−⇀2h2(g)+o2(g) proceeds at a certain temperature until the partial...
Questions



Business, 21.11.2019 20:31


English, 21.11.2019 20:31

English, 21.11.2019 20:31



Physics, 21.11.2019 20:31



History, 21.11.2019 20:31

History, 21.11.2019 20:31


Mathematics, 21.11.2019 20:31

Social Studies, 21.11.2019 20:31



Mathematics, 21.11.2019 20:31

English, 21.11.2019 20:31

 is the value of the equilibrium constant at this temperature.
is the value of the equilibrium constant at this temperature.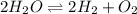
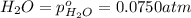
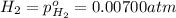
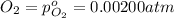
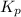 for the given chemical equation is:
for the given chemical equation is: