
You have two 466.0 ml aqueous solutions. solution a is a solution of silver nitrate, and solution b is a solution of potassium chromate. the masses of the solutes in each of the solutions are the same. when the solutions are added together, a blood-red precipitate forms. after the reaction has gone to completion, you dry the solid and find that it has a mass of 331.8 g. (a) calculate the concentration of the potassium ions in the original potassium chromate solution.(b) calculate the concentration of the chromate ions in the final solution

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 18:00
How is the composition of a meteorite relevant to finding out the composition of earth's core?
Answers: 3

Chemistry, 22.06.2019 07:30
Label a-f based on the table using c for concentrated and d for dilute
Answers: 2

Chemistry, 22.06.2019 13:50
Abeaker with 2.00×102 ml of an acetic acid buffer with a ph of 5.000 is sitting on a benchtop. the total molarity of acid and conjugate base in this buffer is 0.100 m. a student adds 4.70 ml of a 0.360 m hcl solution to the beaker. how much will the ph change? the pka of acetic acid is 4.740.
Answers: 1

Chemistry, 22.06.2019 19:30
Astudent conducts an experiment to determine how the amount of water given to a plant affects its growth. what is the independent variable for this experiment?
Answers: 1
You know the right answer?
You have two 466.0 ml aqueous solutions. solution a is a solution of silver nitrate, and solution b...
Questions


Advanced Placement (AP), 18.11.2020 17:10

Mathematics, 18.11.2020 17:10





History, 18.11.2020 17:10

World Languages, 18.11.2020 17:10

Mathematics, 18.11.2020 17:10


English, 18.11.2020 17:10







Mathematics, 18.11.2020 17:10

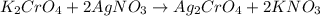
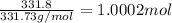
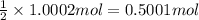 of silver nitrate.
of silver nitrate.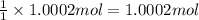 of potassium chromate
of potassium chromate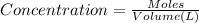
![[K^+]=\frac{2.0004 mol}{0.466 L}=4.2927 mol/L](/tpl/images/0127/0717/630d0.png)
![[CrO_4^{2+}]=\frac{1.0002 mol}{0.466 L+0.466L}=1.0731 mol/L](/tpl/images/0127/0717/e3c96.png)


