
Chemistry, 23.07.2019 06:20 rainbowboy9231
Agaseous compound is 78.14 percent boron and 21.86 percent hydrogen. at 27°c, 74.3 ml of the gas exerted a pressure of 1.12 atm. if the mass of the gas was 0.0934 g, what is its molecular formula?

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 01:00
How can you use chemical equations to predict the products of the reaction you can carry out?
Answers: 1

Chemistry, 22.06.2019 08:30
What is the independent variable in this investigation? mass volume sample number substance density
Answers: 3

Chemistry, 22.06.2019 12:00
What is the lowest number energy level where a d sublevel is found
Answers: 1
You know the right answer?
Agaseous compound is 78.14 percent boron and 21.86 percent hydrogen. at 27°c, 74.3 ml of the gas exe...
Questions

History, 30.09.2019 18:30



Business, 30.09.2019 18:30

Mathematics, 30.09.2019 18:30

Social Studies, 30.09.2019 18:30

History, 30.09.2019 18:30


Chemistry, 30.09.2019 18:30

Mathematics, 30.09.2019 18:30

Mathematics, 30.09.2019 18:30

English, 30.09.2019 18:30

Mathematics, 30.09.2019 18:30

Physics, 30.09.2019 18:30

Mathematics, 30.09.2019 18:30


Mathematics, 30.09.2019 18:30



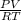
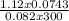
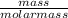
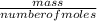
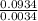
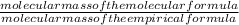
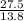 = 2
= 2

