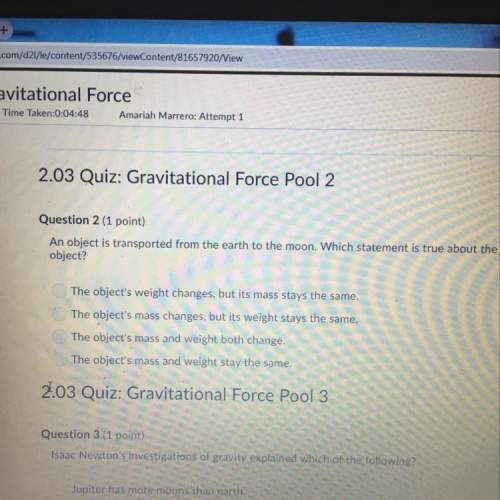
In an ionic compound, the size of the ions affects the internuclear distance (the distance between the centers of adjacent ions), which affects lattice energy (a measure of the force needed to pull ions apart). the lattice energy affects the enthalpy of solution, which can affect solubility. based on ion sizes, rank these compounds by their expected solubilities in water.
rbl
rbbr
rbcl
rbf

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:00
How much would the freezing point of water decrease if 4 mol of sugar were added to 1 kg of water(k=1.86 c/mol/kg for water and i=1 for sugar
Answers: 1

Chemistry, 22.06.2019 09:20
Give the orbital configuration of the phosphorus (p) atom.
Answers: 1

Chemistry, 22.06.2019 16:00
He table below gives the atomic mass and relative abundance values for the three isotopes of element m. relative abundance (%) atomic mass (amu) 78.99 23.9850 10.00 24.9858 11.01 25.9826 what is the average atomic mass (in amu) of element m? 2.86 5.36 24.30 24.98
Answers: 2

Chemistry, 22.06.2019 17:30
Which scenario is most similar to the type of collision that gas particles have according to kinetic molecular theory
Answers: 1
You know the right answer?
In an ionic compound, the size of the ions affects the internuclear distance (the distance between t...
Questions


Mathematics, 09.02.2021 14:00

History, 09.02.2021 14:00

Mathematics, 09.02.2021 14:00


Social Studies, 09.02.2021 14:00

Mathematics, 09.02.2021 14:00

Biology, 09.02.2021 14:00


Mathematics, 09.02.2021 14:00

Computers and Technology, 09.02.2021 14:00


Mathematics, 09.02.2021 14:00

Mathematics, 09.02.2021 14:00

English, 09.02.2021 14:00

Mathematics, 09.02.2021 14:00


Biology, 09.02.2021 14:00

Computers and Technology, 09.02.2021 14:00

Advanced Placement (AP), 09.02.2021 14:00