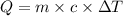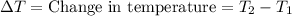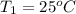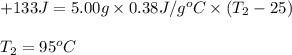Chemistry, 19.07.2019 01:20 katelynn73
A5.00-g sample of copper metal at 25.0 °c is heated by the addition of 133 j of energy. the final temperature of the copper is °c. the specific heat capacity of copper is 0.38 j/g°c.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:00
What mass of carbon dioxide is produced from the complete combustion of 4.50×10−3 g of methane?
Answers: 2

Chemistry, 22.06.2019 12:30
Consider the four elements above. which one of these elements will combine with oxygen in a 1: 1 ratio?
Answers: 3

Chemistry, 22.06.2019 14:00
650.j is the same amount of energy as? 2720cal1550cal650.cal2.72cal
Answers: 2

Chemistry, 22.06.2019 15:00
‘which reaction would most likely require the use of an inert electrode?
Answers: 1
You know the right answer?
A5.00-g sample of copper metal at 25.0 °c is heated by the addition of 133 j of energy. the final te...
Questions

Physics, 16.09.2020 22:01

Mathematics, 16.09.2020 22:01

Mathematics, 16.09.2020 22:01

Mathematics, 16.09.2020 22:01

Mathematics, 16.09.2020 22:01

Mathematics, 16.09.2020 22:01

Mathematics, 16.09.2020 22:01

Spanish, 16.09.2020 22:01

Mathematics, 16.09.2020 22:01

English, 16.09.2020 22:01

Mathematics, 16.09.2020 22:01

Mathematics, 16.09.2020 22:01

English, 16.09.2020 22:01

Mathematics, 16.09.2020 22:01

Social Studies, 16.09.2020 22:01

Health, 16.09.2020 22:01

Mathematics, 16.09.2020 22:01

Mathematics, 16.09.2020 22:01

Social Studies, 16.09.2020 22:01

Mathematics, 16.09.2020 22:01