
The bronsted-lowry definition of acids and bases refers to the transfer of a "proton" from the acid to the base; however, the symbol for a proton (p+) is not generally used in this context. what is the chemical symbol that is commonly used to represent a "proton" in the context of bronsted-lowry acids and bases?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 10:20
In a reaction equation, where are the products located? a.) above the arrow b.) to the right of the arrow c.) to the left of the arrow d.) below the arrow
Answers: 2

Chemistry, 22.06.2019 12:00
What is the subscript for oxygen in its molecular formula
Answers: 1


Chemistry, 22.06.2019 13:30
What does the xylem do? stores the glucose captures the sunlight absorbs oxygen into the leaf carries water from the roots to the leaves
Answers: 1
You know the right answer?
The bronsted-lowry definition of acids and bases refers to the transfer of a "proton" from the acid...
Questions



Engineering, 01.03.2021 17:00

Mathematics, 01.03.2021 17:00

Physics, 01.03.2021 17:00

Advanced Placement (AP), 01.03.2021 17:00

Mathematics, 01.03.2021 17:00

English, 01.03.2021 17:00






Mathematics, 01.03.2021 17:10




Mathematics, 01.03.2021 17:10


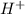
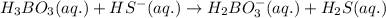
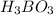 is loosing a proton, thus it is considered as an acid and after losing a proton, it forms
is loosing a proton, thus it is considered as an acid and after losing a proton, it forms 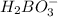 which is a conjugate base.
which is a conjugate base.
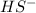 is gaining a proton, thus it is considered as a base and after gaining a proton, it forms
is gaining a proton, thus it is considered as a base and after gaining a proton, it forms 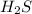 which is a conjugate acid.
which is a conjugate acid.

