
Chemistry, 11.07.2019 22:30 IsabellaGracie
For real gases, how does a change in pressure affect the ratio of pvto nrt?
a. the ratio increases as pressure increases.
b. the ratio is constant at all pressures.
c. the ratio decreases as pressure increases.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 23:30
For the following dehydrohalogenation (e2) reaction, draw the zaitsev product(s) resulting from elimination involving c3–c4 (i.e., the carbon atoms depicted with stereobonds). show the product stereochemistry clearly. if there is more than one organic product, both products may be drawn in the same box. ignore elimination involving c3 or c4 and any carbon atom other than c4 or c3.
Answers: 3


Chemistry, 22.06.2019 14:30
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2

Chemistry, 22.06.2019 15:30
Which of the following are correct values for the ideal gas laws constant r
Answers: 1
You know the right answer?
For real gases, how does a change in pressure affect the ratio of pvto nrt?
a. the ratio incr...
a. the ratio incr...
Questions



English, 29.08.2019 19:10

Social Studies, 29.08.2019 19:10















Computers and Technology, 29.08.2019 19:10

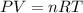
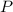 is the pressure of the gas
,
is the pressure of the gas
,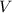 is the volume of gas
,
is the volume of gas
,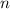 is the number of moles
,
is the number of moles
,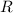 is the gas constant; and
is the gas constant; and 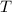 is the temperature of the gas.
is the temperature of the gas.

