
Chemistry, 06.07.2019 03:20 yazmineespinozarive
Determine the amount of heat (in kj) given off when 1.26 × 104 g of ammonia are produced according to the equation n2(g) + 3h2(g) ⟶ 2nh3(g) δh°rxn = −92.6 kj/mol assume that the reaction takes place under standardstate conditions at 25°c.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:00
Match the name of the following compound: mgso4 · h2omagnesium sulfate monohydratemagnesium (ii) sulfate monohydratemagnesium (ii) sulfate hydratemagnesium sulfate hydrate
Answers: 1

Chemistry, 22.06.2019 09:00
Chemical energy is a form of a. kinetic energy only. b. both potential and kinetic energy. c. neither potential nor kinetic energy. d. potential energy only. reset
Answers: 1

Chemistry, 22.06.2019 16:00
How will the volume of a gas be affected if the pressure is tripled, but the temperature remains the same?
Answers: 3

Chemistry, 22.06.2019 23:30
Substance a is a nonpolar liquid and has only dispersion forces among its constituent particles. substance b is also a nonpolar liquid and has about the same magnitude of dispersion forces among its constituent particles. when substance a and b are combined, they spontaneously mix.
Answers: 1
You know the right answer?
Determine the amount of heat (in kj) given off when 1.26 × 104 g of ammonia are produced according t...
Questions

Biology, 05.05.2021 04:10

Mathematics, 05.05.2021 04:10

Chemistry, 05.05.2021 04:10

English, 05.05.2021 04:10



Mathematics, 05.05.2021 04:10

Mathematics, 05.05.2021 04:10


Mathematics, 05.05.2021 04:10

Biology, 05.05.2021 04:10

Mathematics, 05.05.2021 04:10

History, 05.05.2021 04:10

Physics, 05.05.2021 04:10

Mathematics, 05.05.2021 04:10

Mathematics, 05.05.2021 04:10



Mathematics, 05.05.2021 04:10

Computers and Technology, 05.05.2021 04:10

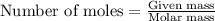
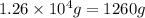
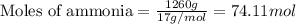
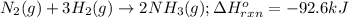
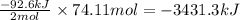 of energy.
of energy.

