The equilibrium constant, kc, for the reaction
n2o4(g)? 2no2(g)
is 4.9×10? 3.
if t...

Chemistry, 05.07.2019 20:20 ronaldhernandez598
The equilibrium constant, kc, for the reaction
n2o4(g)? 2no2(g)
is 4.9×10? 3.
if the equilibrium mixture contains [no2] = 0.050 m , what is the molar concentration of n2o4?
express the concentration to two significant figures and include the appropriate units.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:10
56.16 gregor mendel was the first scientist to use statistics to analyze scientific data. before mendel's experiments, scientists believed that organisms acquired traits from their environment and passed them on to their offspring. after mendel's discoveries were accepted, scientists realized that traits passed to offspring were the result of genes being passed from parents to offspring. this is an example of pls
Answers: 1

Chemistry, 22.06.2019 08:40
For each of the following compounds, write the formula then predict whether it would be a strong, weak, or non-electrolyte when placed in di water. for the ionic compounds only, put (s) or (aq) after the forrmula formula strong, weak or non electrolyte? a calcium hydroxide b. silver carbonate c. lead(ii) sulfate d. phosphorus trifluoride e. sodium phosphide f barium sulfate g. strontium acetate h. zinc nitrate
Answers: 3

Chemistry, 22.06.2019 10:30
Skills of homo sapiens were found an excavation. the skulls were preserved because the bodies were frozen. so, these fossils are (blank) fossils.the image shows the evolution of skulls beginning 2 to 3 million years ago. based on the image, modern human skulls(blank) ape skulls.
Answers: 1

Chemistry, 22.06.2019 23:10
Amines are good nucleophiles, even though they are neutral molecules. how would the rate of an sn2 reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? amines are good nucleophiles, even though they are neutral molecules. how would the rate of an reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? because both reactants in the rate-limiting step are neutral, the reaction will be faster if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will be slower if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will occur at the same rate if the polarity of the solvent is increased. request answer
Answers: 3
You know the right answer?
Questions


Mathematics, 18.11.2020 04:30

Mathematics, 18.11.2020 04:30

Mathematics, 18.11.2020 04:30

Arts, 18.11.2020 04:30

History, 18.11.2020 04:30

History, 18.11.2020 04:40


Mathematics, 18.11.2020 04:40




Computers and Technology, 18.11.2020 04:40


Biology, 18.11.2020 04:40

Mathematics, 18.11.2020 04:40



History, 18.11.2020 04:40

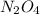 is,
is, 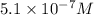

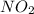 = 0.050 M
= 0.050 M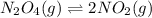
![K_c=\frac{[NO_2]^2}{[N_2O_4]}](/tpl/images/0055/3008/271f5.png)
![4.9\times 10^3=\frac{(0.050)^2}{[N_2O_4]}](/tpl/images/0055/3008/a5c3c.png)
![[N_2O_4]=5.1\times 10^{-7}M](/tpl/images/0055/3008/c9f7d.png)



