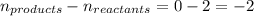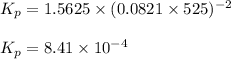Chemistry, 05.07.2019 18:20 aliceohern
Methanol can be synthesized from monoxide and hydrogen gas at 525 k. a reaction mixture consisting initially of 1.8 moles of co and 2.2 moles of h2 in 5.0-l container was found to contain 0.6 moles of ch3oh after reaching equilibrium (a) calculate equilibrium concentration (in molarity) of co and h2 (b) calculate equilibrium constants kc and kp for this reaction

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:00
During chemistry class, carl performed several lab tests on two white solids. the results of three tests are seen in the data table. based on this data, carl has concluded that substance b must have bonds.
Answers: 2

Chemistry, 22.06.2019 06:30
What effect might melting sea ice have for people who live in coastal areas?
Answers: 1

Chemistry, 23.06.2019 00:50
50 points! need answer asap. what type of organic compound contains the following functional group? (2 points)
Answers: 3

You know the right answer?
Methanol can be synthesized from monoxide and hydrogen gas at 525 k. a reaction mixture consisting i...
Questions



Chemistry, 20.09.2021 16:10

Mathematics, 20.09.2021 16:10








Chemistry, 20.09.2021 16:10


Social Studies, 20.09.2021 16:10

Physics, 20.09.2021 16:10





Mathematics, 20.09.2021 16:10

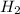 are 0.24 M and 0.32 M.
are 0.24 M and 0.32 M. are 1.5625 and
are 1.5625 and 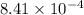
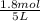
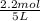
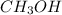 = 0.6
= 0.6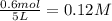
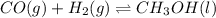
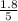
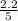 0
0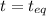
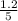
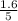
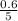
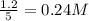
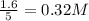
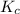 for the given chemical reaction follows:
for the given chemical reaction follows:![K_c=\frac{[CH_3OH]}{[CO][H_2]}](/tpl/images/0054/9747/63b2d.png)
![[CH_3OH]=0.12mol/L](/tpl/images/0054/9747/f965f.png)
![[CO]=0.24mol/L](/tpl/images/0054/9747/ee0b0.png)
![[H_2]=0.32mol/L](/tpl/images/0054/9747/aa037.png)
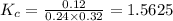
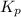 with
with 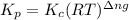
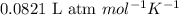
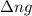 = change in number of moles of gas particles =
= change in number of moles of gas particles =