
Chemistry, 02.07.2019 19:10 silviamgarcia
At 35°c, kc = 1.6 multiplied by10-5 for the following reaction
2 nocl(g) reverse reaction arrow 2 no(g)+ cl2(g)
calculate the concentrations of all species at equilibrium if
2.0 mol no and 1.0 mol of cl2 are placed in a 1.0 l flask

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:40
Three alkali metals in group 1 are a. calcium, strontium, barium b. boron, aluminum, gallium c. sodium, potassium, rubidium d. fluorine, iodine, chlorine
Answers: 1

Chemistry, 22.06.2019 19:00
How many liters of ethylene glycol antifreeze (c2h6o2), with a density of 1.100 g/l, would you add to your car radiator containing 15.0 kg of water if you needed to protect your engine to - 21.5°c? for water, kf = 1.86°c m -1.
Answers: 1

Chemistry, 22.06.2019 20:20
The characteristics of two different types of reactions are shown below: reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of an element. which statement is true about the atoms of the elements that participate in the two reactions? their identity changes in both reaction a and reaction b. their identity changes in reaction a but not in reaction b. their identity changes in reaction b but not in reaction a. their identity remains the same in both reaction a and reaction b.
Answers: 1

Chemistry, 22.06.2019 20:30
Which states of matter have particles that move independently of one another with very little attraction?
Answers: 1
You know the right answer?
At 35°c, kc = 1.6 multiplied by10-5 for the following reaction
2 nocl(g) reverse reaction arro...
2 nocl(g) reverse reaction arro...
Questions


Computers and Technology, 24.09.2020 14:01

Mathematics, 24.09.2020 14:01


Mathematics, 24.09.2020 14:01






History, 24.09.2020 14:01

Biology, 24.09.2020 14:01

Mathematics, 24.09.2020 14:01

Mathematics, 24.09.2020 14:01

Engineering, 24.09.2020 14:01



Mathematics, 24.09.2020 14:01

Mathematics, 24.09.2020 14:01

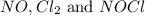 are, 0.05 M, 0.043 M and 0.975 M respectively.
are, 0.05 M, 0.043 M and 0.975 M respectively.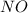 = 2 mole
= 2 mole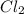 = 1 mole
= 1 mole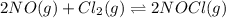
![K_c=\frac{[NOCl]^2}{[NO]^2[Cl_2]}](/tpl/images/0043/6110/56950.png)
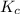 for reverse reaction =
for reverse reaction = 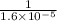
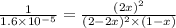
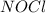 = x M = 0.975 M
= x M = 0.975 M


