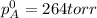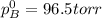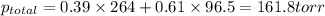Chemistry, 02.07.2019 04:10 NikkiZoeller
Liquid a has a vapor pressure of 264 torr at 20∘c, and liquid b has a vapor pressure of 96.5 torr at the same temperature. if 5.50 moles of liquid a and 8.50 moles of liquid b are combined to form an ideal solution, what is the total vapor pressure (in torr) above the solution at 20.0∘c?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Ibeg i need 20. a reaction produces 4.93 l of oxygen, but was supposed to produce 1 mol of oxygen. what is the percent yield?
Answers: 1

Chemistry, 22.06.2019 03:30
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1

Chemistry, 22.06.2019 10:00
What is the atomic mass of an atom that has 6 protons, 6 neutrons, and 6 electrons? a) 6 b) 8 c) + 1 d) 12 e) 18
Answers: 1

You know the right answer?
Liquid a has a vapor pressure of 264 torr at 20∘c, and liquid b has a vapor pressure of 96.5 torr at...
Questions

Geography, 15.07.2019 16:30



Arts, 15.07.2019 16:30

Physics, 15.07.2019 16:30

Health, 15.07.2019 16:30

Chemistry, 15.07.2019 16:30

History, 15.07.2019 16:30

Arts, 15.07.2019 16:30

Computers and Technology, 15.07.2019 16:30

Chemistry, 15.07.2019 16:30

Computers and Technology, 15.07.2019 16:30

Social Studies, 15.07.2019 16:30

Health, 15.07.2019 16:30

History, 15.07.2019 16:30


History, 15.07.2019 16:30


History, 15.07.2019 16:30

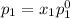 and
and 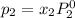
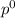 = pressure in the pure state
= pressure in the pure state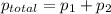
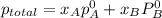
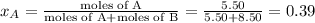 ,
, 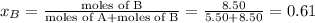 ,
,