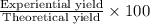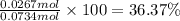Ammonia, nh3nh3 , can react with oxygen to form nitrogen gas and water. 4nh3(aq)+3o2(g)⟶2n2(g)+6h2o(l) 4nh3(aq)+3o2(g)⟶2n2(g)+6h2o(l) if 2.35 g2.35 g nh3nh3 reacts with 3.53 g3.53 g o2o2 and produces 0.650 l0.650 l n2n2 , at 295 k295 k and 1.01 bar1.01 bar , which reactant is limiting? o2(g)o2(g) nh3(aq)nh3(aq) calculate the percent yield of the reaction. percent yield:

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 11:40
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3

Chemistry, 22.06.2019 15:20
Identify arrows pointing to bonding electrons. done h-0-0-h ) intro
Answers: 3

Chemistry, 22.06.2019 22:30
What is a number added in front of a formula in order to balance the equation
Answers: 1
You know the right answer?
Ammonia, nh3nh3 , can react with oxygen to form nitrogen gas and water. 4nh3(aq)+3o2(g)⟶2n2(g)+6h2o(...
Questions


History, 20.11.2019 15:31



History, 20.11.2019 15:31





Mathematics, 20.11.2019 15:31

History, 20.11.2019 15:31

Health, 20.11.2019 15:31


Mathematics, 20.11.2019 15:31



Mathematics, 20.11.2019 15:31

History, 20.11.2019 15:31


Health, 20.11.2019 15:31

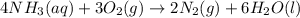
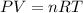
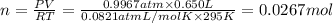
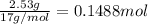
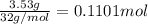
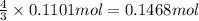 of ammonia.
of ammonia.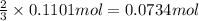 of nitrogen.
of nitrogen.