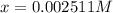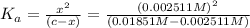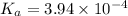Chemistry, 28.06.2019 21:30 tilievaughn14
Aspirin (acetylsalicylic acid, c9h8o4) is a weak monoprotic acid. to determine its acid-dissociation constant, a student dissolved 2.00 g of aspirin in 0.600 l of water and measured the ph. what was the ka value calculated by the student if the ph of the solution was 2.60?

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 04:20
Which of the following is true for the actual yield of a reaction? it is always calculated as a ratio. it is the yield from the excess reactant. it is the yield from the limiting reactant. it is always less than the theoretical yield.
Answers: 1

Chemistry, 22.06.2019 08:00
Will give ! what are the advantages and disadvantages of nuclear power? check all that apply. one advantage of nuclear energy is that it does not produce carbon dioxide emissions. storage of nuclear waste is a short-term problem associated with nuclear energy. the problem with uranium mining is that a large quantity of uranium must be extracted to meet energy needs because the energy release from uranium fission is so low. safe operation of a nuclear power plant can be jeopardized by a human mistake.
Answers: 1

Chemistry, 22.06.2019 17:30
In a heat of an engine, if 700 j enters the system, and the piston does 400 j of work what is the final internal (thermal) energy of the system if the initial energy is 1,500 j
Answers: 2
You know the right answer?
Aspirin (acetylsalicylic acid, c9h8o4) is a weak monoprotic acid. to determine its acid-dissociation...
Questions

English, 06.07.2019 15:50

Mathematics, 06.07.2019 15:50


Business, 06.07.2019 15:50

Mathematics, 06.07.2019 15:50

History, 06.07.2019 15:50

Mathematics, 06.07.2019 15:50








Mathematics, 06.07.2019 15:50

Chemistry, 06.07.2019 15:50


Mathematics, 06.07.2019 15:50

Mathematics, 06.07.2019 15:50

 .
.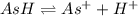
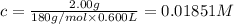
![K_a=\frac{[H^+][As^+]}{[AsH]}=\frac{x\times c}{(c-x)}=\frac{x^2}{(c-x)}](/tpl/images/0028/5803/345f7.png) ..(1)
..(1)![pH=2.60=\log[H^+]=-\log[x]](/tpl/images/0028/5803/39c79.png)