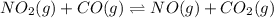Chemistry, 28.06.2019 21:20 tamikalove78
Consider the following reaction at equilibrium: no2(g) + co(g) = no(g) + co2(g) suppose the volume of the system is decreased at constant temperature, what change will this cause in the system? a shift to produce more no a shift to produce more co a shift to produce more no2 no shift will occur

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
The diagram shows the structures of horse and cat forelimbs. what does the diagram suggest about the evolutionary relationship between these two mammals? a. they have homologous structures, indicating a common ancestor. b. they have analogous structures, indicating a common ancestor. c. they have homologous structures, indicating that they do not have a common ancestor. d. they have analogous structures, indicating that they do not have a common ancestor.
Answers: 2

Chemistry, 22.06.2019 08:30
What are the first three quantum numbers for the electrons located in subshell 2s?
Answers: 2

Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2

Chemistry, 22.06.2019 22:30
You just calculated that the heat of fusion for chloromethane is 6400 j/mol. the heat of fusion for hydrogen is 120 j/mol.? which of the following account for this difference? more than one correcta. chloromethane can absorb more energy at the same temperature. b. hydrogen has stronger intermolecular forces than chloromethane. c. hydrogen molecules can pack more closely than chloromethane molecules. d. chloromethane experiences dipole-dipole interactions. e. chloromethane has a higher molar mass than hydrogen.
Answers: 3
You know the right answer?
Consider the following reaction at equilibrium: no2(g) + co(g) = no(g) + co2(g) suppose the volume...
Questions






Mathematics, 03.05.2021 17:50

Mathematics, 03.05.2021 17:50


Computers and Technology, 03.05.2021 17:50


History, 03.05.2021 17:50

Geography, 03.05.2021 17:50


Mathematics, 03.05.2021 17:50


Computers and Technology, 03.05.2021 18:00

History, 03.05.2021 18:00


Mathematics, 03.05.2021 18:00

Mathematics, 03.05.2021 18:00