
Chemistry, 26.06.2019 16:10 pchisholm100
Aluminum chloride can be formed from its elements:
(i) 2al(s) + 3cl2 (g) ⟶ 2alcl3 (s) δh° = ? use the reactions here to determine the δh° for reaction (i):
(ii) hcl(g) ⟶ hcl(aq) δh(ii) ° = −74.8 kj
(iii) h2 (g) + cl2 (g) ⟶ 2hcl(g) δh(iii) ° = −185 kj
(iv) alcl3 (aq) ⟶ alcl3 (s) δh(iv) ° = +323 kj/mol
(v) 2al(s) + 6hcl(aq) ⟶ 2alcl3 (aq) + 3h2 (g) δh(v) ° = −1049 kj

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
200. ml of 3.00 m nacl solution is diluted to a final volume of 500. ml. what is the molarity of the final solution?
Answers: 2

Chemistry, 22.06.2019 03:10
Agas diffuses 1/7 times faster than hydrogen gas (h2). what is the molar mass of the gas? 100.10 g/mol 98.78 g/mol 86.68 g/mol 79.98 g/mol
Answers: 3

Chemistry, 22.06.2019 17:30
Air can be considered a mixture. which statement does not explain why?
Answers: 1

Chemistry, 22.06.2019 23:30
Why do oxygen have a strong attractive force for electrons
Answers: 2
You know the right answer?
Aluminum chloride can be formed from its elements:
(i) 2al(s) + 3cl2 (g) ⟶ 2alcl3 (s) δh° =...
(i) 2al(s) + 3cl2 (g) ⟶ 2alcl3 (s) δh° =...
Questions

Social Studies, 16.02.2021 19:00

Mathematics, 16.02.2021 19:00

Mathematics, 16.02.2021 19:00


Mathematics, 16.02.2021 19:00

Mathematics, 16.02.2021 19:00


English, 16.02.2021 19:00

Advanced Placement (AP), 16.02.2021 19:00

Social Studies, 16.02.2021 19:00

Physics, 16.02.2021 19:00



Mathematics, 16.02.2021 19:00

Mathematics, 16.02.2021 19:00

Mathematics, 16.02.2021 19:00

Social Studies, 16.02.2021 19:00



Business, 16.02.2021 19:00

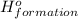 for the reaction is -1406.8 kJ.
for the reaction is -1406.8 kJ. is:
is:
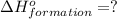
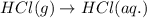
 ( × 6)
( × 6)
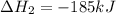 ( × 3)
( × 3)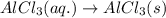
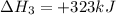 ( × 2)
( × 2)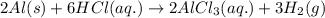
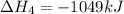
![\Delta H^o_{formation}=[6\times \Delta H_1]+[3\times \Delta H_2]+[2\times \Delta H_3]+[1\times \Delta H_4]](/tpl/images/0020/0754/79ecd.png)
![\Delta H^o_{formation}=[(-74.8\times 6)+(-185\times 3)+(323\times 2)+(-1049\times 1)]=-1406.8kJ](/tpl/images/0020/0754/37adf.png)


