
Chemistry, 26.06.2019 05:20 sindy35111
The combustion of ethane (c2h6)(c2h6) produces carbon dioxide and steam. 2c2h6(g)+7o2(g)⟶4co2(g)+6h2o(g) 2c2h6(g)+7o2(g)⟶4co2(g)+6h2o(g) how many moles of co2co2 are produced when 5.95 mol5.95 mol of ethane is burned in an excess of oxygen? moles of co2: co2:

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Plz choose one of the compounds from the table and explain how you know the numbers of atoms in your formula. is it possible for two different compounds to be made from the exact same two elements? why or why not? with a limited number of elements (less than 120 are known), does this mean we also have a small number of compounds or do we have a large number of compounds in this world?
Answers: 1

Chemistry, 22.06.2019 02:40
How many liters of hydrogen gas will be produced at stp from the reaction of 7.179×10^23 atoms of magnesium with 54.219g of phosphoric acid (h3po4) the equation is 3mg + 2h3(> mg(po4)2+3h2
Answers: 1


Chemistry, 22.06.2019 13:00
Lab reagent, hypothesis test.a reference solution used as a lab reagent is purported to have a concentration of 5 mg/dl. six samples are taken from this solution and the following concentrations are recorded: (5.32, 4.88, 5.10, 4.73, 5.15, 4.75) mg/dl.these six measurements are assumed to be an srs of all possible measurements from solution.they are also assumed to have a standard deviation of 0.2, a normal distributin, and a mean concentration equal to the true concentration of the solution.carry out a significance test to determine whether these six measurements provide reliable evidence that the true concentration of the solution is actually not 5 mg/dl.
Answers: 1
You know the right answer?
The combustion of ethane (c2h6)(c2h6) produces carbon dioxide and steam. 2c2h6(g)+7o2(g)⟶4co2(g)+6h2...
Questions

English, 03.01.2020 21:31

Social Studies, 03.01.2020 21:31

Social Studies, 03.01.2020 21:31


Mathematics, 03.01.2020 21:31




English, 03.01.2020 21:31



Social Studies, 03.01.2020 21:31




Mathematics, 03.01.2020 21:31

Mathematics, 03.01.2020 21:31

Chemistry, 03.01.2020 21:31

Social Studies, 03.01.2020 21:31

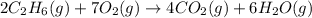
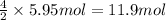 of carbon-dioxide
of carbon-dioxide

