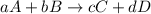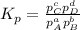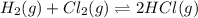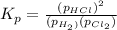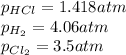Chemistry, 26.06.2019 03:20 elopezhilario6339
At a given temperature, 4.06 atm of h2 and 3.5 atm of cl2 are mixed and allowed to come to equilibrium. the equilibrium pressure of hcl is found to be 1.418 atm. calculate kp for the reaction at this temperature. h2(g) + cl2(g) < => 2 hcl(g)

Answers: 1
Another question on Chemistry

Chemistry, 20.06.2019 18:04
Can anyone (a-level) a student was analysing a carbonate compound, mco3, containing an unknown group 2 metal, m. the student carried out thermal decomposition on 0.730g of the carbonate and measured the volume of gas produced. mco3 (s) mo (s) + co2 (g) the student collected and measured 120cm3 of carbon dioxide. 1 mol of carbon dioxide occupies 24 000 cm3 under these conditions. calculate the molar mass of the group 2 carbonate and hence deduce the identity of the group 2 metal, m.
Answers: 3

Chemistry, 22.06.2019 03:30
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1

Chemistry, 22.06.2019 06:30
Identify the missing numbers below to show the result of multiplying the numbers (1.6 × 10-19)(5.0 × 106) = c × 10d
Answers: 1

Chemistry, 22.06.2019 09:10
When a nucleus absorbs a neutron and then breaks apart, there are many products of the reaction. what is not a product of a nuclear fission reaction
Answers: 1
You know the right answer?
At a given temperature, 4.06 atm of h2 and 3.5 atm of cl2 are mixed and allowed to come to equilibri...
Questions



Mathematics, 19.12.2021 17:30



English, 19.12.2021 17:30

Chemistry, 19.12.2021 17:30

Mathematics, 19.12.2021 17:30

Mathematics, 19.12.2021 17:30


Biology, 19.12.2021 17:30







Spanish, 19.12.2021 17:40

Mathematics, 19.12.2021 17:40

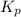 for the given chemical reaction is 0.1415
for the given chemical reaction is 0.1415