
Chemistry, 21.06.2019 16:20 maevemboucher78
Aluminum reacts with chlorine gas to form aluminum chloride via the following reaction: 2al(s)+3cl2(g)→2alcl3(s) what is the maximum mass of aluminum chloride that can be formed when reacting 32.0 g of aluminum with 37.0 g of chlorine? express your answer to three significant figures and include the appropriate units.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 16:30
Asample of silver (with work function ? = 4.52 ev) is exposed to an ultraviolet light source (? = 200 nm), which results in the ejection of photoelectrons. what changes will be observed if: silver is replaced with copper (? = 5.10 ev) more photoelectrons ejected no photoelectrons are emitted fewer photoelectrons ejected more energetic photoelectrons (on average) less energetic photoelectrons (on average)
Answers: 3

Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1

Chemistry, 22.06.2019 09:20
What will most likely happen when two bromine atoms bond together?
Answers: 3

Chemistry, 22.06.2019 13:30
Which of the following has wavelength longer than the wavelength of viable light? a) x rays b) gamma rays c) radios waves d) ultraviolet waves
Answers: 1
You know the right answer?
Aluminum reacts with chlorine gas to form aluminum chloride via the following reaction: 2al(s)+3cl2...
Questions

French, 18.10.2019 07:00


Biology, 18.10.2019 07:00







History, 18.10.2019 07:00

Business, 18.10.2019 07:00

History, 18.10.2019 07:00


Chemistry, 18.10.2019 07:00

English, 18.10.2019 07:00

Mathematics, 18.10.2019 07:00




 ....(1)
....(1) 
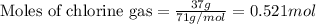
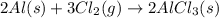
 of aluminium.
of aluminium.


