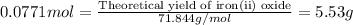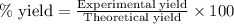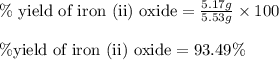Chemistry, 26.10.2019 11:43 Kathryn014
For the following reaction, 4.31 grams of iron are mixed with excess oxygen gas . the reaction yields 5.17 grams of iron(ii) oxide . iron ( s ) + oxygen ( g ) iron(ii) oxide ( s ) what is the theoretical yield of iron(ii) oxide ? 21.6 grams what is the percent yield for this reaction ? 85 %

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:40
Consider the nuclear equation below. 239/94 pu—-> x+ 4/2 he. what is x?
Answers: 2

Chemistry, 22.06.2019 09:20
What will most likely happen when two bromine atoms bond together?
Answers: 3

Chemistry, 22.06.2019 12:50
The number at the end of an isotope’s name is the number.
Answers: 1

Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3
You know the right answer?
For the following reaction, 4.31 grams of iron are mixed with excess oxygen gas . the reaction yield...
Questions

Physics, 01.08.2019 22:00


Physics, 01.08.2019 22:00




Computers and Technology, 01.08.2019 22:00







History, 01.08.2019 22:00

Mathematics, 01.08.2019 22:00

Biology, 01.08.2019 22:00

English, 01.08.2019 22:00


Health, 01.08.2019 22:00

History, 01.08.2019 22:00

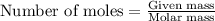 ....(1)
....(1)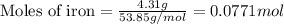
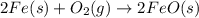
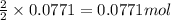 of iron (ii) oxide
of iron (ii) oxide