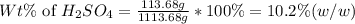Chemistry, 22.06.2019 02:10 fvmousdiana
Determine the percent sulfuric acid by mass of a 1.61 m aqueous solution of h2so4. %

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 15:30
Count the number of each type of atom in the equation below, and then balance the equation. write in the numbers of atoms and coefficients. add a 1 if there should be no coefficient. cs2(l) + o2(g) → co2(g) + so2(g) c [ ] s [ ] o > c [ ] s [ ] o [ ] cs2(l) + [ ] o2(g) > [ ] co2(g) + [ ] so2(g)
Answers: 3

Chemistry, 22.06.2019 16:50
Answer asap need it by wednesday morning calculate the ph of 0.02m hcl best answer will be brainliest
Answers: 1

Chemistry, 22.06.2019 23:00
The data below were determined for the reaction shown below. s2o82– + 3i – (aq) → 2so42– + i3– expt. # [s2o82–] (m) [i –] (m) initial rate 1 0.038 0.060 1.4 × 10 – 5 m/s 2 0.076 0.060 2.8 × 10 – 5 m/s 3 0.076 0.030 1.4 × 10 – 5 m/s the rate law for this reaction must be:
Answers: 1

Chemistry, 23.06.2019 02:00
Calculate the molarity of each aqueous solution: a. 78.0 ml of 0.240 m naoh diluted to 0.250 l with water b. 38.5 ml of 1.2 m hno3 diluted to 0.130 l with water
Answers: 1
You know the right answer?
Determine the percent sulfuric acid by mass of a 1.61 m aqueous solution of h2so4. %...
Questions

Biology, 13.07.2019 16:00


History, 13.07.2019 16:00

Mathematics, 13.07.2019 16:00


Social Studies, 13.07.2019 16:00


Mathematics, 13.07.2019 16:00

History, 13.07.2019 16:00

Social Studies, 13.07.2019 16:00


Chemistry, 13.07.2019 16:00

Chemistry, 13.07.2019 16:00

Mathematics, 13.07.2019 16:00


Geography, 13.07.2019 16:00

Social Studies, 13.07.2019 16:00

English, 13.07.2019 16:00


Health, 13.07.2019 16:00