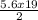Chemistry, 22.06.2019 20:30 Schoolworkspace453
Consider the following unbalanced equation for the combustion of hexane: αc6h14(g)+βo2(g)→γco2(g)+δh2o(g) part a balance the equation. give your answer as an ordered set of numbers α, β, γ, use the least possible integers for the coefficients. α α , β, γ, δ = nothing request answer part b determine how many moles of o2 are required to react completely with 5.6 moles c6h14. express your answer using two significant figures. n n = nothing mol request answer provide feedback

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 14:00
3. if the dartboard below is used to model an atom, which subatomic particles would be located at z?
Answers: 2

Chemistry, 22.06.2019 04:30
How many grams of co(g) are there in 74.5 ml of the gas at 0.933 atm and 30o c?
Answers: 1

Chemistry, 22.06.2019 09:00
Chemical energy is a form of a. kinetic energy only. b. both potential and kinetic energy. c. neither potential nor kinetic energy. d. potential energy only. reset
Answers: 1

Chemistry, 22.06.2019 13:40
Can someone me with 6 to 10 plz this is for masteries test.
Answers: 1
You know the right answer?
Consider the following unbalanced equation for the combustion of hexane: αc6h14(g)+βo2(g)→γco2(g)+δ...
Questions

Mathematics, 16.01.2020 08:31

English, 16.01.2020 08:31

Mathematics, 16.01.2020 08:31

Mathematics, 16.01.2020 08:31

Mathematics, 16.01.2020 08:31


Mathematics, 16.01.2020 08:31




Mathematics, 16.01.2020 08:31

History, 16.01.2020 08:31

Mathematics, 16.01.2020 08:31





English, 16.01.2020 08:31

English, 16.01.2020 08:31

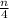 )O₂ → xCO₂ +
)O₂ → xCO₂ + 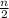 H₂O
H₂O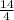 = 6 + 3.5 = 9
= 6 + 3.5 = 9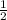
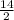 = 7
= 7