
Chemistry, 23.06.2019 01:30 jonmorton159
At a certain temperature the rate of this reaction is first order in hi with a rate constant of : 0.0632s2hig=h2g+i2g suppose a vessel contains hi at a concentration of 1.28m . calculate how long it takes for the concentration of hi to decrease to 17.0% of its initial value. you may assume no other reaction is important. round your answer to 2 significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:00
If there is any 12 to 14 girls that need a boyfriend just follow me and let me know
Answers: 1

Chemistry, 22.06.2019 07:30
In a reaction (at equilibrium) that makes more moles of gas than it consumes, what is the effect of increasing the pressure?
Answers: 1

Chemistry, 22.06.2019 12:00
A5.000 g sample of niso4 h2o decomposed to give 2.755 g of anhydrous niso4. what is the formula of the hydrate? what is the full chemical name for the hydrate? what is the molar mass of the hydrate? niso4•_h2o what is the mass % of water in the hydrate?
Answers: 1

Chemistry, 22.06.2019 15:20
Water is initially present in a state where its molecules are far apart. during a change of state, its molecules slow down. which change of state has most likely taken place? from a gas to a liquid from a liquid to a gas from a solid to a liquid from a gas to a plasma
Answers: 1
You know the right answer?
At a certain temperature the rate of this reaction is first order in hi with a rate constant of : 0....
Questions

Advanced Placement (AP), 16.04.2021 23:20


Mathematics, 16.04.2021 23:20

Mathematics, 16.04.2021 23:20



Arts, 16.04.2021 23:20

SAT, 16.04.2021 23:20

English, 16.04.2021 23:20

History, 16.04.2021 23:20

Health, 16.04.2021 23:20

Mathematics, 16.04.2021 23:20

Mathematics, 16.04.2021 23:20

Mathematics, 16.04.2021 23:20



History, 16.04.2021 23:20



Biology, 16.04.2021 23:20

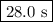
![\ln \left (\dfrac{[A]_{0}}{[A]} \right ) = kt](/tpl/images/0006/1343/aeb04.png)
![\begin{array}{rcl}\ln \left (\dfrac{[A]_{0}}{0.170[A]_{0}} \right ) & = & 0.0632t\\\\\ln \left (5.882) & = & 0.0632t\\1.772 & = & 0.0632t\\\\t & = & \dfrac{1.772}{0.0632}\\\\t & = & \textbf{{28.0 s}}\\\end{array}\\\text{It will take } \boxed{\textbf{28.0 s}} \text{ for [HI] to decrease to 17.0 \% of its original value.}](/tpl/images/0006/1343/cc68a.png)


