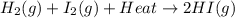The reaction below shows a system in equilibrium. how would a decrease in temperature affect this reaction? a. the rate of formation of the gases would increase. b. the equilibrium of the reaction would shift to the left. c. the equilibrium would shift to cause the gases to sublime into solids. d. the chemicals on the left would quickly form the chemical on the right.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 19:20
What is the strongest intermolecular force between an nacl unit and an h2o molecule together in a solution? covalent bonding dipole-dipole force hydrogen bonding ion-dipole force
Answers: 1

Chemistry, 22.06.2019 19:00
Structure of the atoms: discovery of the nucleus in 1909i need answering all of these questions
Answers: 3

Chemistry, 23.06.2019 00:30
•hydration •dissociation •dissolving which one goes to which
Answers: 1

You know the right answer?
The reaction below shows a system in equilibrium. how would a decrease in temperature affect this r...
Questions


Health, 18.09.2019 14:00



Chemistry, 18.09.2019 14:00




Chemistry, 18.09.2019 14:00

Biology, 18.09.2019 14:00


History, 18.09.2019 14:00

Arts, 18.09.2019 14:00

English, 18.09.2019 14:00




Biology, 18.09.2019 14:00

Geography, 18.09.2019 14:00