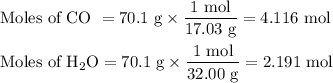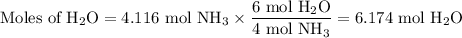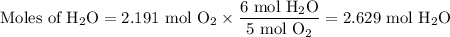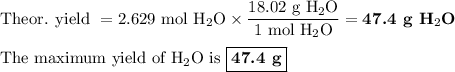The ostwald process is used commercially to produce nitric acid, which is, in turn, used in many modern chemical processes. in the first step of the ostwald process, ammonia is reacted with oxygen gas to produce nitric oxide and water. what is the maximum mass of h2oh2o that can be produced by combining 70.170.1 g of each reactant?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:30
This element exists in adundance in the sun.explain how you would go about capturing sunlight.would this captured sunlight contain any of the element?
Answers: 1

Chemistry, 22.06.2019 02:30
Asa choose the correct set of reaction coefficients to properly balance the following chemical equation according to the law of conservation of mass: __s8 + __o2 ==> __so2 1, 1, 8 1, 8, 1 1, 8, 8 8, 1, 1
Answers: 1

Chemistry, 23.06.2019 02:30
what is your question? collegechemistry 5+3 pts in november 1987, a massive iceberg broke loose from the antartic ice mass and floated free in the ocean. the chunk of ice was estimated to be 98 mi long, 25 mi wide, and 750 ft thick. a typical backyard swimming pool contains about 24,000 gallons of water. how many of these pools could you fill from the water in this iceberg? (assume the iceberg is a rectangular solid of the above dimensions and consists of water only). express answer in scientific notation.
Answers: 3

You know the right answer?
The ostwald process is used commercially to produce nitric acid, which is, in turn, used in many mod...
Questions

Advanced Placement (AP), 25.02.2021 14:00

Geography, 25.02.2021 14:00

Mathematics, 25.02.2021 14:00



Computers and Technology, 25.02.2021 14:00



Business, 25.02.2021 14:00







Mathematics, 25.02.2021 14:00


English, 25.02.2021 14:00

Physics, 25.02.2021 14:00

Mathematics, 25.02.2021 14:00