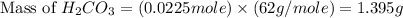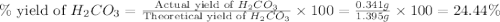Chemistry, 25.06.2019 17:10 sweetbri7p5v6tn
During a laboratory experiment, a 3.81-gram sample of nahco3 was thermally decomposed. in this experiment, carbon dioxide and water vapors escape and are combined to form carbonic acid. after decomposition, the sample weighed 2.86 grams. calculate the percentage yield of carbonic acid for the reaction. describe the calculation process in detail. nahco3 → na2co3 + h2co3

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:30
Two atoms interact with each other as shown by the equation. complete the equation by filling in the missing parts. 1 2 3 4 5 h he li
Answers: 2

Chemistry, 22.06.2019 13:10
Which electron configuration represents the electrons in an atom of sodium in the ground state at stp
Answers: 1

Chemistry, 22.06.2019 16:00
Sulfuric acid is a polyprotic acid. write balanced chemical equations for the sequence of reactions that sulfuric acid can undergo when it's dissolved in water.
Answers: 2

Chemistry, 22.06.2019 19:20
The equation picture below shows which type of nuclear reaction u 235 + n x e 134 + sr 100 + 2n
Answers: 1
You know the right answer?
During a laboratory experiment, a 3.81-gram sample of nahco3 was thermally decomposed. in this exper...
Questions

Mathematics, 07.01.2020 11:31


Social Studies, 07.01.2020 11:31

Mathematics, 07.01.2020 11:31

English, 07.01.2020 11:31

Mathematics, 07.01.2020 11:31

Mathematics, 07.01.2020 11:31

History, 07.01.2020 11:31

Mathematics, 07.01.2020 11:31






Mathematics, 07.01.2020 11:31



Social Studies, 07.01.2020 11:31


Mathematics, 07.01.2020 11:31

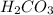 is, 24.44 %
is, 24.44 %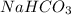 .
.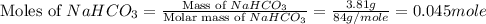
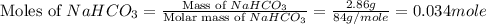
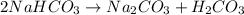
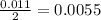 mole of
mole of 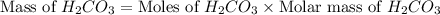
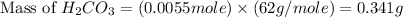
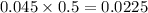 mole of
mole of