Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 12:50
Use the standard enthalpies of formation for the reactants and products to solve for the δhrxn for the following reaction. (the δhf of c2h4 is 52.26 kj/mol, co2 is -393.509 kj/mol, and h2o is -241.818 kj.) c2h4 (g) + 3o2(g) 2co2 (g) + 2h2o(g) δhrxn = the reaction is .
Answers: 3

Chemistry, 22.06.2019 00:30
This is a characteristic of the elements in the periodic table that shows a pattern. it may increase or decrease across or down the table.
Answers: 1

Chemistry, 23.06.2019 02:00
What are fossils of organisms that existed over a wide area but only for a limited time period called?
Answers: 2

Chemistry, 23.06.2019 04:00
Achemical reaction is done in the setup shown , resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 2
You know the right answer?
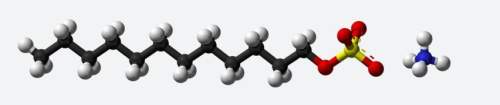
(98 ) ill mark brainliestdescribe the structure of ammonium lauryl sulfate. refer to the given diagr...
Questions


History, 27.09.2019 20:20

Health, 27.09.2019 20:20


Mathematics, 27.09.2019 20:20






Biology, 27.09.2019 20:20

Mathematics, 27.09.2019 20:20



English, 27.09.2019 20:20

Mathematics, 27.09.2019 20:20

Mathematics, 27.09.2019 20:20


History, 27.09.2019 20:20