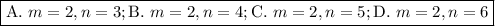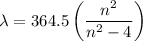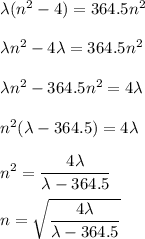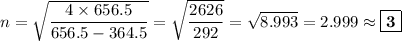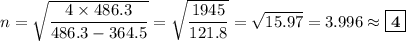Determine the balmer formula n and m values for the wavelength 656.5 nm. possible choices: m= 1 n= 2 m= 2 n= 3 m= 3 n= 4 m= 2 n= 5 part b determine the balmer formula n and m values for the wavelength 486.3 nm. possible choices: m= 1 n=2 m= 2 n=3 m= 1 n=4 m= 2 n=4 part c determine the balmer formula n and m values for the wavelength 434.2 nm. possible choices: m= 1 n= 4 m= 2 n= 4 m= 3 n= 4 m= 2 n= 5 part d determine the balmer formula n and m values for the wavelength 410.3 nm. possible choices: m= 2 n= 4 m= 2 n= 5 m= 3 n= 4 m= 2 n= 6

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:20
Describing intermolecular forces use the drop down menus to match the type of intermolecular force to its name dipole dipole interactions dipole induced dipole interactions london dispersion forces hydrogen bond van der waals forces done
Answers: 1

Chemistry, 22.06.2019 13:30
An animal cell loses the ability to convert energy stored in food to energy that the cell can use. which of the cell's organelles has stopped working? a.the mitochondria b.the nucleus c.the vacuoles d.the endoplasmic reticulum
Answers: 1

Chemistry, 22.06.2019 14:00
The two naturally occurring isotopes of chlorine are 35cl (34.969 amu, 75.77%) and 37cl (36.966 amu, 24.23%). the two naturally occurring isotopes of bromine are 79br (78.918 rm amu, 50.69%) and 81br (80.916 amu, 49.31%). chlorine and bromine combine to form bromine monochloride, brcl. 1. how many peaks will be present in a mass spectrum for brcl? the four combinations of molecule possible given these four isotopes are: 81br37cl, 81br35cl, 79br37cl, and 79br35cl. 2. what are the masses of the four different brcl molecules? express the masses using six significant figures, in decreasing numeric order (highest to lowest), separated by commas.
Answers: 3

Chemistry, 22.06.2019 20:00
Glucose (c6h12o6) is an important biological molecule. (round the answer to nearest hundredth.) what is the percent by mass of carbon in glucose?
Answers: 2
You know the right answer?
Determine the balmer formula n and m values for the wavelength 656.5 nm. possible choices: m= 1 n=...
Questions

Mathematics, 17.09.2020 17:01

Mathematics, 17.09.2020 17:01

Mathematics, 17.09.2020 17:01

History, 17.09.2020 17:01

Mathematics, 17.09.2020 17:01

English, 17.09.2020 17:01

Mathematics, 17.09.2020 17:01

Mathematics, 17.09.2020 17:01

Mathematics, 17.09.2020 17:01

Mathematics, 17.09.2020 17:01

Mathematics, 17.09.2020 17:01

Mathematics, 17.09.2020 17:01

Mathematics, 17.09.2020 17:01

Mathematics, 17.09.2020 18:01

Mathematics, 17.09.2020 18:01

Mathematics, 17.09.2020 18:01

Mathematics, 17.09.2020 18:01

Mathematics, 17.09.2020 18:01

Mathematics, 17.09.2020 18:01

Mathematics, 17.09.2020 18:01