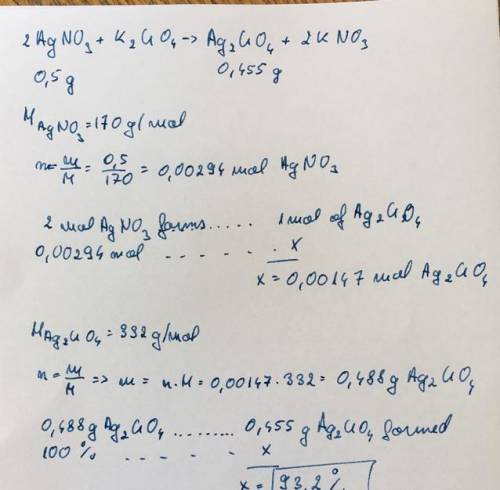Chemistry, 27.06.2019 07:20 maxi12312345
3. calculate the percent yield of silver chromate if 0.455 grams of silver chromate are producedfrom 0.500 grams of silver nitrate.2 agno3 + k2cro4 -> ag2 cro 4 + 2 kno3

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Select the correct answer. given: 2libr + ba → babr2 + 2li in this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced? a. 1.18 mol b. 2.37 mol c. 4.73 mol d. 16.4 mol e. 32.9 mol
Answers: 2


Chemistry, 22.06.2019 20:30
Which of the following is not true about the atomic model of substances?
Answers: 1

You know the right answer?
3. calculate the percent yield of silver chromate if 0.455 grams of silver chromate are producedfrom...
Questions

Biology, 19.07.2019 08:30


English, 19.07.2019 08:30

Spanish, 19.07.2019 08:30

Physics, 19.07.2019 08:30











History, 19.07.2019 08:30