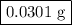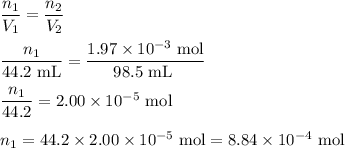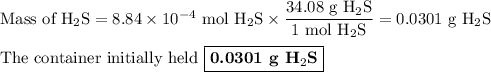Chemistry, 28.06.2019 05:50 dinarussell74
Hydrogen sulfide gas (h2s) is a highly toxic gas that is responsible for the smell of rotten eggs. the volume of a container of hydrogen sulfide is 44.2ml. after the addition of more hydrogen sulfide, the volume increases to 98.5ml under constant pressure and temperature. the container now holds 1.97×10−3mol of the gas. how many grams of hydrogen sulfide were in the container initially? give your answer in three significant figures.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 16:30
How many grams of mgbr2 are needed to produce 75g or metal?
Answers: 1

Chemistry, 22.06.2019 18:30
Which of the following words describe the reality that the universe looks the same from various perspective
Answers: 3

Chemistry, 22.06.2019 22:30
What methods could you use to solubilize calcium carbonate
Answers: 1

Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1
You know the right answer?
Hydrogen sulfide gas (h2s) is a highly toxic gas that is responsible for the smell of rotten eggs. t...
Questions

History, 30.11.2021 02:50






History, 30.11.2021 02:50

Mathematics, 30.11.2021 02:50


Computers and Technology, 30.11.2021 02:50

Business, 30.11.2021 02:50


Mathematics, 30.11.2021 02:50


SAT, 30.11.2021 02:50

SAT, 30.11.2021 02:50

English, 30.11.2021 02:50

Mathematics, 30.11.2021 02:50

Mathematics, 30.11.2021 02:50