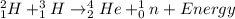Chemistry, 29.06.2019 07:10 kirbymichellrow6zq9
Each reation releases or absorbs a very large amount of energy per atom. a. chemical and nuclear reactionb. chemical reactions. c. no reaction d. nuclear reation

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
What are the charges of the subatomic particles by choosing the answer from the drop down menu. protons have a (+1,0,or-1). (protons,neutrons,electrons) have a 0 charge. 3.) electrons have a (+1,0,-1)
Answers: 2

Chemistry, 22.06.2019 15:30
Which of the following are correct values for the ideal gas laws constant r
Answers: 1

Chemistry, 22.06.2019 16:10
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2

Chemistry, 23.06.2019 10:30
Fill in the blanks for the following statements: the rms speed of the molecules in a sample of h2 gas at 300 k will be times larger than the rms speed of o2 molecules at the same temperature, and the ratio µrms (h2) / µrms (o2) with increasing temperature. a not enough information is given to answer this question b sixteen, will not change c four, will not change d four, will increase e sixteen, will decrease
Answers: 2
You know the right answer?
Each reation releases or absorbs a very large amount of energy per atom. a. chemical and nuclear rea...
Questions

Mathematics, 05.10.2020 01:01

Mathematics, 05.10.2020 01:01

Biology, 05.10.2020 01:01

Mathematics, 05.10.2020 01:01

Engineering, 05.10.2020 01:01



Business, 05.10.2020 01:01



English, 05.10.2020 01:01


Mathematics, 05.10.2020 01:01

Mathematics, 05.10.2020 01:01


Biology, 05.10.2020 01:01

Computers and Technology, 05.10.2020 01:01


Biology, 05.10.2020 01:01

Physics, 05.10.2020 01:01